A newly released analysis from the Ethics and Public Policy Center (EPPC) suggests that removing federal safety requirements for the abortion pill has increased risks for women, particularly when the drug is prescribed without an in-person medical visit.
The report, written by EPPC President Ryan T. Anderson and Director of Data Analysis Jamie Bryan Hall, examines adverse event data for the abortion drug mifepristone before and after the U.S. Food and Drug Administration (FDA) eliminated its longstanding requirement that the drug be dispensed during an in-person medical appointment. This new analysis builds on the largest-known study of the abortion pill, which Dr. Anderson and Hall coauthored and published last year.
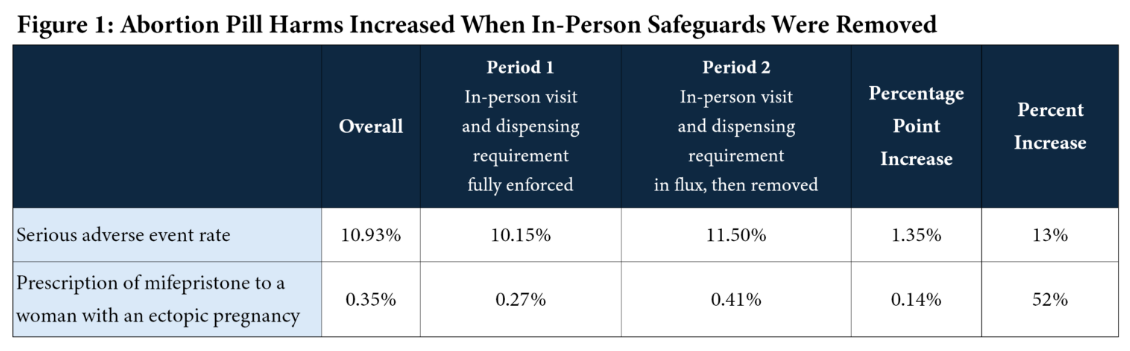
According to the analysis, the rate of serious complications following chemical abortion increased after the rule change. Researchers found that serious adverse events rose from 10.15 percent when the in-person requirement was enforced to 11.50 percent after the requirement was removed, a statistically significant increase.
[Click here to subscribe to Pregnancy Help News!]
The study also found that prescriptions for the abortion pill given to women experiencing ectopic pregnancy—a potentially life-threatening condition—rose significantly during the same period. Researchers reported that the rate increased from 0.27 percent before the policy change to 0.41 percent afterward, a rise of more than 50 percent.
For two decades after the abortion pill was approved in 2000, the FDA required women to receive the drug during an in-person visit with a medical professional. The visit allowed clinicians to confirm gestational age, rule out ectopic pregnancy, and assess whether the woman was being coerced.
In 2020, during the COVID-19 pandemic, the FDA temporarily suspended the in-person requirement. The agency later made the change permanent, allowing prescriptions to be issued through telemedicine and mailed directly to patients.
Ryan T. Anderson, president of the Ethics and Public Policy Center, and Jamie Bryan Hall, the organization’s director of data analysis, authored the new report analyzing prescription data from January 2017 through December 2023.
The researchers compared outcomes before July 2020—when the in-person requirement was still in place—with outcomes afterward.
While the study found an overall rise in complications, the authors suggest the impact of remote prescribing could be significantly higher than the overall numbers indicate.
Because the exact share of abortion pills dispensed remotely is not fully documented, researchers modeled several scenarios. If remote prescriptions accounted for 10 percent of mifepristone prescriptions, the analysis estimates the serious adverse event rate for those cases could reach 23.65 percent, more than double the rate associated with in-person dispensing.
In a second scenario where remote prescriptions represented 25 percent of cases, the projected serious adverse event rate would be 15.55 percent, still substantially higher than in-person care.
The report also warns that diagnosing ectopic pregnancy is difficult without an ultrasound or in-person examination. As a result, the analysis suggests the abortion pill could be prescribed to women with ectopic pregnancies three to six times more often when dispensed remotely.
Chemical abortion now accounts for the majority of abortions in the United States. According to the Guttmacher Institute, a think tank previously affiliated with Planned Parenthood, 63 percent of abortions were performed using abortion pills in 2023, with telehealth and mail-order prescriptions increasing rapidly in recent years.
The authors argue that the rise of online abortion services has made it difficult to ensure patient safety.
Tweet This: The rise of online abortion services has made it difficult to ensure patient safety. @RyanTAnd @Mr_Jamie_B_Hall @EPPCdc
“In-person medical visits play an important role in determining gestational age, identifying ectopic pregnancy, and confirming that a woman is not being coerced,” Anderson and Hall wrote in their analysis.
For several years, pregnancy help organizations and other national pro-life groups have urged federal officials to restore the earlier safety standards for the abortion pill.
Leaders in the pregnancy help movement have argued that the removal of the in-person requirement reduced medical oversight at a time when chemical abortion is increasingly obtained through telehealth and mail-order services. They say basic safeguards, such as in-person medical screening and accurate drug labeling, are necessary to protect women’s health and ensure that serious conditions like ectopic pregnancy are properly diagnosed.
The EPPC report adds new data to those concerns, suggesting that the removal of the in-person dispensing rule corresponds with measurable increases in complications.
The findings come amid an ongoing national debate over abortion pill regulations.
Supporters of expanded telehealth access argue that mail-order prescriptions increase access to abortion, especially in states with strong pro-life laws. However, many note that removing in-person safeguards reduces medical oversight and puts women at greater risk for insufficient care and abuse.
Researchers conclude that restoring the earlier FDA safety requirements—particularly the in-person medical visit—would help address the increased risks highlighted in their analysis. They also note that many voters support stronger safety protocols. Polling cited by the authors suggests a large majority of Americans favor requiring an in-person doctor visit before prescribing abortion pills and updating warning labels to reflect real-world safety risks. Even Americans who disagree about abortion itself support safeguards intended to protect women’s health.